If a Substance Is Corrosive What Do You Know About the Substance
Corrosive substances can pose a serious adventure to the workplace. Every bit a subversive substance, information technology's of import to empathise the hazardous properties of corrosive substances, so yous can ensure they're used and stored in a rubber , compliant manner. Corrosive substances are defined as materials that can attack and destroy, by chemical action, living tissue, organic compounds and metallic. Recognised equally "Grade eight Corrosive Substances" in The Australian Dangerous Appurtenances Lawmaking (ADG Code), they accept the adequacy of causing astringent harm to people, property and the environment. These corrosive chemicals are highly chancy when they are inhaled, swallowed or come into contact with the eyes or skin. Exposure to these materials tin can upshot in the called-for of human tissue, which tin can cause severe scarring, lung bug and even incomprehension. Severe exposure to corrosive substances can be fatal. And while we are all familiar with everyday examples of corrosion such as rusty atomic number 26, corroded batteries or rusted out cars, corrosive substances tin chop-chop impairment or destroy belongings, send and other goods that they come up into contact. Even a pocket-sized leak or spill of a corrosive substance requires immediate attention to forbid the destruction of belongings and the contamination of the immediate environment. So how can you properly manage the handling and storage of corrosive substances in your workplace? First, it's important to understand the properties of the substance so y'all can formulate the right strategy to ensure information technology'southward safely used and independent. And one of the key measures of a liquid'due south corrosive backdrop is its pH level. Ranked on a scale betwixt 1 and xiv, pH is a measurement of how acidic or basic a water-soluble substance is. If a substance is considered neutral, such as pure h2o, it volition have a pH level of 7. As the substance becomes increasingly acidic, the pH level decreases from seven. For example, lemon juice has a pH level of 2, coffee may have a pH level of 5 and rainwater has a value of 5.five. Annihilation with a pH level to a higher place 7 indicates that the substance is alkali metal. For example, some paw soap may take a pH level of 10, while bleach may have a pH level of 12. Again, as the pH value increases, the alkaline level also increases. A pH of 4 is ten times more acidic that a pH of 5, and 100 times more acidic than a pH of half-dozen. Likewise, a pH of 13 is 10 times more alkaline than a pH of 12, and 100 times more than alkaline metal than a pH of xi. Therefore, the more acidic or more alkaline a substance is, the more than effective it will be as a corrosive substance. Acids have a pH level lower than 7, while bases have a pH level higher than vii. If a substance is deemed equally highly corrosive, it must exist stored in compliant storage facilities such equally a corrosive storage cabinet . These purpose-built cabinets will finer minimise the hazards that corrosive substances pose upon your workplace, such as dangerous vapours and spills. But what are some examples of ordinarily used acids and bases? We will now talk over some examples of commonly used acids and bases. Hydrochloric Acid Hydrochloric acid (HCL) has a pH level of 1.1 at 38% concentration. If this acid accidently came into contact with your optics or peel, it would immediately begin to dissolve your peel tissue. HCL is commonly used in the product of batteries, fireworks, leather and building materials. It is besides usually used in water and table salt purification. Your body too produces Hydrochloric acrid to assistance your digestive system. Sulfuric Acid Sulfuric acid (H 2 Then iv ) has a pH of 0.5 at a concentration of 33.v%, which is equivalent to the concentration of sulfuric acid used in lead-acrid batteries. Sulfuric acid is one of the almost important industrial chemicals. It plays a part in the production of a lot of manufactured appurtenances. Despite existence primarily used in the production of fertilizers such as superphosphates, sulfates and ammonium phosphates, sulfuric acid is also widely used in the production of dyes, paints, fabric pigments, explosives, lubricants, metals and batteries. Sulfuric acid is a dehydrating agent and information technology reacts violently with water. When in contact with water, sulfuric acid will boil and splash. If sulfuric acid is released into the atmosphere, it tin can cause acrid rain which is very harmful to the surroundings. Nitric Acid Nitric acid (HNO 3 ) has a pH level of ane.ii at standard commercial concentration of 68%. The uses of Nitric acid are very similar to those of sulfuric acid. Nitric acid is used in the production of ammonium nitrate, plastics, dyes and explosives such as nitroglycerin and TNT. When nitric acrid is combined with HCL, a fuming liquid, known equally aqua regia is formed capable of dissolving gold and platinum. Nitric acid is also utilised in the medical industry to remove warts and every bit a colorimetric test to distinguish drugs. When exposed to the skin, nitric acrid tin crusade astringent burns, ulceration, dermatitis and yellow staining. As a corrosive substance, nitric acrid is capable of dissolving many materials including most metals. For ultimate prophylactic nitric needs to be stored in a compliant safety cabinet to prevent the risk of acrid burns and damage to expensive property, institute and equipment. Chromic Acrid Chromic acid (H 2 CrO 4 ) has a pH level of approximately 3.03. It is used as a cleaning agent and in the manufacturing of glazes, colored glass and chromium plating. In the past, it has commonly been used equally a bleach to dye pilus. Due to the high bear on chromic acids have on the environment and human health, it'south no longer used for this purpose. Again, chromic acrid will burn your skin and safe handling and storage is crucial. Acetic Acid Acetic acid (CH 3 COOH) or equally we more than commonly know it, vinegar, has a pH of ii.4 at 5% dilute solution. Acetic acid is primarily used in the production of vinyl acetate monomer, followed past ester production and used to create solvents for inks, paints and coatings. Acerb acid has proved to be widely useful in the medical industry, especially in the handling of cancer amongst many other conditions such as ear infections. And of course, acetic acid is used in food production. Surprisingly enough, vinegar is only 4-xviii% acetic acid by mass. Despite seeming reasonably harmless, acetic acid, specifically in full-bodied amounts can be considerably harmful to human wellness and the surroundings. Ammonium Hydroxide Ammonium hydroxide (NH 4 OH) has a pH of ten.09 and is frequently used as a glass cleaning agent in various commercial and industrial products. Y'all will too find ammonium hydroxide in food items where it is used as a food condiment to the correct level of acerbity. This corrosive base is also ordinarily used as a refrigerant and can be found in a range of products including detergents, textiles, soaps, ceramics, pharmaceuticals, inks and explosives. As ammonium hydroxide may crusade extensive tissue damage if it is exposed to the skin, it must be stored in a prophylactic and compliant manner . Potassium Hydroxide Potassium hydroxide (KOH) has a pH of 10.98 and is commonly used in the product of fertilisers, biodiesels and soft soaps. Potassium hydroxide is used as an electrolyte in a process usually referred to as chemical cremation or resomation. Sodium Hydroxide Sodium Hydroxide (NaOH) has a pH of xiii. Although it is predominantly used in the paper industry, information technology plays an important part in tissue digestion and in the manufacturing of sodium salts and detergents. Sodium hydroxide tin can also exist found in cleaning agents and every bit a pH regulator in organic synthesis and metallic production. If sodium hydroxide is not handled and stored in a safe corrosive storage cabinet , it tin can exist dangerous to human health and the environment. Corrosive Substance Storage Cabinet - 250L Sodium Hypochlorite Sodium hypochlorite (NaClO) has a pH of approximately 13 at 10-15% concentration. It is used in diverse industries, including waste management, every bit a bleach or disinfectant. Sodium hypochlorite also has other applications, including as an antiseptic in the medical industry and as a pesticide in the agricultural industry. From mining operations to hospitality venues, a diverse range of industries regularly use corrosive substances at their workplaces. Now that you have a amend understanding of corrosive substances and their pH levels, it's important to consider proven strategies that will minimise their harm, including the safe storage of corrosive substances. One important attribute of safe storage is ensuring that course 8 corrosive substances are segregated from other incompatible classes of dangerous goods. If you'd like to learn how to segregate corrosive substances from other incompatible classes of dangerous appurtenances, download our free dangerous appurtenances segregation nautical chart past clicking on the epitome below.  Corroded Metallic
Corroded Metallic What is a pH Level?
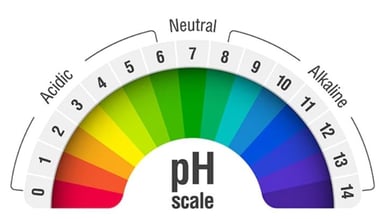 pH Scale
pH Scale What Are Some Examples Of Corrosive Acids?
 Corrosive Acid
Corrosive Acid Some Examples Of Corrosive Bases

Storing Corrosive Chemicals & Substances In The Workplace
Source: https://blog.storemasta.com.au/examples-corrosive-substances-ph-levels
0 Response to "If a Substance Is Corrosive What Do You Know About the Substance"
إرسال تعليق